Abstract
Review Article
Advances in the use of GABAergic interneurons for the treatment of epilepsy
Giorgio R Merlo*, Federica Frisina#, Giulia Valetti#, Giulia Zuccarini# and Luciano Conti#
Published: 04 September, 2019 | Volume 3 - Issue 1 | Pages: 009-022
Forebrain GABAergic neurons, the main inhibitory type of neuron in the cortex and hippocampus, represent a highly heterogeneous cell population that has been implicated in the predisposition to epilepsy and the onset of seizure. Earlier attempts to restore inhibition and reduce seizure in animal models of epilepsy have been carried out using embryonic basal forebrain tissue as source of immature GABAergic progenitors in cell-based therapies, with promising results. For therapeutic strategies this approach appears unrealistic, while the use of pluripotent stem cells to obtain immature GABAergic neurons opens new and promising avenues. Research on neural stem cells and pluripotent stem cells has greatly advanced and protocols have been established to efficiently direct progenitor cells to differentiate towards the GABAergic lineage. However, being highly heterogeneous, these neurons are difficult to be fully represented in vitro. Better knowledge on the expressed gene profiles, at single cell level, and the differentiation trajectory of these neurons will consent a more precise monitoring of the differentiation steps. Here we review the current literature about how to obtain and characterize genuine inhibitory neurons, how these can be grafted in animal models (and one day possibly in human) and which diseases could potentially be targeted and the efficiency of therapeutic outcome. The main obstacles that need to be overcome are: a) choice of an appropriate animal model, b) availability of human cells prone to GABA differentiation, c) the full representation of all IN subtypes, their proportions and their physiological activities, d) how to monitor them on the long-term after transplant.
Read Full Article HTML DOI: 10.29328/journal.jsctt.1001014 Cite this Article Read Full Article PDF
Keywords:
Neural stem cells; Neuronal differentiation; Inhibitory neurons; Epilepsy; Cell therapy
References
- Chang BS, Lowenstein DH. Epilepsy. N Engl J Med. 2003; 349: 1257-1266.
- Galanopoulou AS. Mutations affecting GABAergic signaling in seizures and epilepsy. Pflüg Arch. Eur J Physiol. 2010; 460: 505-523. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/20352446
- Goodwin M. The importance of brand continuity in epilepsy drugs. Nurs Times. 2005; 101: 26-27. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15997944
- Herman S. Intractable Epilepsy: Relapsing, Remitting, or Progressive? Epilepsy Curr. 2010; 10: 146-148. PubMed: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3001022/
- Walia KS, Khan EA, Ko DH, Raza SS, Khan YN. Side Effects of Antiepileptics- A Review. Pain Pract. 2004; 4: 194-203. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/17173601
- Zhu Q, Naegele JR, Chung S. Cortical GABAergic Interneuron/Progenitor Transplantation as a Novel Therapy for Intractable Epilepsy. Front Cell Neurosci. 2018; 12: 167. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/29997478
- Hovinga CA, Asato MR, Manjunath R, Wheless JW, Phelps SJ, et al. Association of non-adherence to antiepileptic drugs and seizures, quality of life, and productivity: Survey of patients with epilepsy and physicians. Epilepsy Behav. 2008; 13: 316-322. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/18472303
- Southwell DG, Nicholas CR, Basbaum AI, Stryker MP, Kriegstein AR, et al. Interneurons from Embryonic Development to Cell-Based Therapy. Science. 2014; 344: 1240622-1240622
- Markram H, Toledo-Rodriguez M, Wang Y, Gupta A, Silberberg G, et al. Interneurons of the neocortical inhibitory system. Nat Rev Neurosci. 2004; 5: 793-807.
- The Petilla Interneuron Nomenclature Group (PING), Ascoli GA, Alonso-Nanclares L, Anderson SA, Barrionuevo G, et al. Petilla terminology: nomenclature of features of GABAergic interneurons of the cerebral cortex. Nat Rev Neurosci. 2008; 9: 557-568. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/18568015
- Tasic B, Menon V, Nguyen TN, Kim TK, Jarsky T, et al. Adult mouse cortical cell taxonomy revealed by single cell transcriptomics. Nat Neurosci. 2016; 19: 335-346. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26727548
- Kelsom C, Lu W. Development and specification of GABAergic cortical interneurons. Cell Biosci. 2013; 3: 19. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26727548
- The Petilla Interneuron Nomenclature Group (PING), Ascoli GA, Alonso-Nanclares L, Anderson SA, Barrionuevo G, et al. Petilla terminology: nomenclature of features of GABAergic interneurons of the cerebral cortex. Nat Rev Neurosci. 2008; 9: 557-568. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/18568015
- Zeisel A, Munoz-Manchado AB, Codeluppi S, Lonnerberg P, La Manno G, et al. Cell types in the mouse cortex and hippocampus revealed by single-cell RNA-seq. Science. 2015; 347: 1138-1142. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/25700174
- Toledo-Rodriguez M, Markram H. Single-cell RT-PCR, a technique to decipher the electrical, anatomical, and genetic determinants of neuronal diversity. Methods Mol Biol. 2014; 1183: 143-158. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/25023306
- Mi D, Li Z, Lim L, Li M, Moissidis M, et al. Early emergence of cortical interneuron diversity in the mouse embryo. Science. 2018; 360: 81-85. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/29472441
- Anderson SA. Interneuron Migration from Basal Forebrain to Neocortex: Dependence on Dlx Genes. Science. 1997; 278: 474-476. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/9334308
- Brunstrom JE, Gray-Swain MR, Osborne PA, Pearlman AL. Neuronal heterotopias in the developing cerebral cortex produced by neurotrophin-4. Neuron. 1997; 18: 505-517. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/9115743
- de Carlos JA, López-Mascaraque L, Valverde F. Dynamics of cell migration from the lateral ganglionic eminence in the rat. J Neurosci. 1996; 16: 6146-6156. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/8815897
- DeDiego I, Smith-Fernández A, Fairén A. Cortical cells that migrate beyond area boundaries: characterization of an early neuronal population in the lower intermediate zone of prenatal rats. Eur J Neurosci. 1994; 6: 983-997. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/7952285
- Tamamaki N, Fujimori KE, Takauji R. Origin and route of tangentially migrating neurons in the developing neocortical intermediate zone. J Neurosci. 1997; 17: 8313-8323. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/9334406
- Butt SJB, Fuccillo M, Nery S, Noctor S, Kriegstein A, et al. The Temporal and Spatial Origins of Cortical Interneurons Predict Their Physiological Subtype. Neuron. 2005; 48: 591-604. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/16301176
- Gelman DM, Martini FJ, Nobrega-Pereira S, Pierani A, Kessaris N, et al. The Embryonic Preoptic Area Is a Novel Source of Cortical GABAergic Interneurons. J Neurosci 2009; 29: 9380-9389. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/19625528
- Xu Q. Origins of Cortical Interneuron Subtypes. J Neurosci. 2004; 24: 2612-2622. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15028753
- Xu Q, Tam M, Anderson SA. Fate mapping Nkx2.1-lineage cells in the mouse telencephalon. J Comp Neurol. 2008; 506: 16-29. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/17990269
- Tanaka DH, Oiwa R, Sasaki E, Nakajima K. Changes in cortical interneuron migration contribute to the evolution of the neocortex. Proc Natl Acad Sci. 2011; 108: 8015-8020.
- Clowry GJ. An enhanced role and expanded developmental origins for gamma-aminobutyric acidergic interneurons in the human cerebral cortex. J Anat. 2015; 227: 384-393. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/24839870
- Hansen DV, Lui JH, Flandin P, Yoshikawa K, Rubenstein JL, et al. Non-epithelial stem cells and cortical interneuron production in the human ganglionic eminences. Nat Neurosci. 2013; 16: 1576-1587. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/24097039
- Al-Jaberi N, Lindsay S, Sarma S, Bayatti N, Clowry GJ. The Early Fetal Development of Human Neocortical GABAergic Interneurons. Cereb Cortex. 2015; 25: 631-645. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/24047602
- Ma T, Wang C, Wang L, Zhou X, Tian M, et al. Subcortical origins of human and monkey neocortical interneurons. Nat Neurosci. 2013; 16: 1588-1597. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/24097041
- Chu K, Kim M, Jung KH, Jeon D, Lee ST, et al. Human neural stem cell transplantation reduces spontaneous recurrent seizures following pilocarpine-induced status epilepticus in adult rats. Brain Res. 2004; 1023: 213-221. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15374747
- Kepecs A, Fishell G. Interneuron cell types are fit to function. Nature. 2014; 505: 318-326. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/24429630
- Klausberger T, Somogyi P. Neuronal Diversity and Temporal Dynamics: The Unity of Hippocampal Circuit Operations. Science. 2008; 321: 53-57. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/18599766
- Jiang X, Shen S, Cadwell CR, Berens P, Sinz F, et al. Principles of connectivity among morphologically defined cell types in adult neocortex. Science. 2015; 350: aac9462-aac9462. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26612957
- Traub RD, Borck C, Colling SB, Jefferys JGR. On the Structure of Ictal Events in Vitro. Epilepsia. 1996; 37: 879-891. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/8814102
- Traub RD, Whittington MA, Colling SB, Buzsáki G, Jefferys JG. Analysis of gamma rhythms in the rat hippocampus in vitro and in vivo. J Physiol. 1996; 493: 471-484. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/8782110
- Curia G, Longo D, Biagini G, Jones RSG, Avoli M. The pilocarpine model of temporal lobe epilepsy. J Neurosci Methods. 2008; 172: 143-157. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/18550176
- Scheffer IE, Berkovic S, Capovilla G, Connolly MB, French J, et al. ILAE classification of the epilepsies: Position paper of the ILAE Commission for Classification and Terminology. Epilepsia. 2017; 58: 512-521. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/28276062
- Avanzini G, Franceschetti S. Cellular biology of epileptogenesis. Lancet Neurol. 2003; 2: 33-42. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/12849299
- Kann O, Kovács R. Mitochondria and neuronal activity. Am J Physiol Cell Physiol 2007; 292: C641-C657. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/17092996
- Papageorgiou IE, Fetani AF, Lewen A, Heinemann U, Kann O. Widespread activation of microglial cells in the hippocampus of chronic epileptic rats correlates only partially with neurodegeneration. Brain Struct Funct. 2015; 220: 2423-2439. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/24878824
- Seifert G, Carmignoto G, Steinhäuser C. Astrocyte dysfunction in epilepsy. Brain Res Rev. 2010; 63: 212-221. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/19883685
- Bissonette GB, Bae MH, Suresh T, Jaffe DE, Powell EM. Prefrontal cognitive deficits in mice with altered cerebral cortical GABAergic interneurons. Behav Brain Res. 2014; 259: 143-151. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/24211452
- Hashemi E, Ariza J, Rogers H, Noctor SC, Martínez-Cerdeño V. The Number of Parvalbumin-Expressing Interneurons Is Decreased in the Prefrontal Cortex in Autism. Cereb Cortex. 2018; 28: 690-690. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26922658
- Inan M, Petros TJ, Anderson SA. Losing your inhibition: linking cortical GABAergic interneurons to schizophrenia. Neurobiol Dis. 2013; 53: 36-48. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/23201207
- Jacob J. Cortical interneuron dysfunction in epilepsy associated with autism spectrum disorders. Epilepsia. 2016; 57: 182-193. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26682992
- Konstantoudaki X, Chalkiadaki K, Tivodar S, Karagogeos D, Sidiropoulou K. Impaired synaptic plasticity in the prefrontal cortex of mice with developmentally decreased number of interneurons. Neuroscience. 2016; 322: 333-345. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26926965
- Ongür D, Prescot AP, McCarthy J, Cohen BM, Renshaw PF. Elevated gamma-aminobutyric acid levels in chronic schizophrenia. Biol. Psychiatry. 2010; 68: 667-670. PubMed: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2942977/
- Takano T. Interneuron Dysfunction in Syndromic Autism: Recent Advances. Dev Neurosci. 2015; 37: 467-475. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26183392
- Yizhar O, Fenno LE, Prigge M, Schneider F, Davidson TJ, et al. Neocortical excitation/inhibition balance in information processing and social dysfunction. Nature. 2011; 477: 171-178. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/21796121
- Yoon JH, Maddock RJ, Rokem A, Silver MA, Minzenberg MJ, et al. GABA concentration is reduced in visual cortex in schizophrenia and correlates with orientation-specific surround suppression. J Neurosci. 2010; 30: 3777-3781. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/20220012
- Marín O. Interneuron dysfunction in psychiatric disorders. Nat Rev Neurosci. 2012; 13: 107-120. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/22251963
- Galanopoulou A. GABAA Receptors in Normal Development and Seizures: Friends or Foes? Curr Neuropharmacol. 2008; 6: 1-20. PubMed: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2645547/
- Galanopoulou AS. Sexually dimorphic expression of KCC2 and GABA function. Epilepsy Res. 2008; 80: 99-113. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/18524541
- Ben-Ari Y. Excitatory actions of gaba during development: the nature of the nurture. Nat Rev Neurosci. 2002; 3: 728-739. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/12209121
- Kato M. Genotype-phenotype correlation in neuronal migration disorders and cortical dysplasias. Front Neurosci. 2015; 9: 181. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26052266
- Cammarota M, Losi G, Chiavegato A, Zonta M, Carmignoto G. Fast spiking interneuron control of seizure propagation in a cortical slice model of focal epilepsy: Fast-spiking interneurons in focal seizure propagation. J Physiol. 2013; 591: 807-822. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/23207591
- Sessolo M, Marcon I, Bovetti S, Losi G, Cammarota M, et al. Parvalbumin-Positive Inhibitory Interneurons Oppose Propagation But Favor Generation of Focal Epileptiform Activity. J Neurosci. 2015; 35: 9544-9557. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26134638
- Shiri Z, Manseau F, Lévesque M, Williams S, Avoli M. Interneuron activity leads to initiation of low-voltage fast-onset seizures: Epileptiform Synchronization. Ann Neurol. 2015; 77: 541-546. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/25546300
- Ye H, Kaszuba S. Inhibitory or excitatory? Optogenetic interrogation of the functional roles of GABAergic interneurons in epileptogenesis. J Biomed Sci. 2017; 24: 93. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/29202749
- Everson JL, Fink DM, Yoon JW, Leslie EJ, Kietzman HW, et al. Sonic hedgehog regulation of Foxf2 promotes cranial neural crest mesenchyme proliferation and is disrupted in cleft lip morphogenesis. Development. 2017; 144: 2082-2091. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/28506991
- Markram H, Toledo-Rodriguez M, Wang Y, Gupta A, Silberberg G, et al. Interneurons of the neocortical inhibitory system. Nat Rev Neurosci. 2004; 5: 793-807. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15378039
- Whittington MA, Traub RD. Interneuron Diversity series: Inhibitory interneurons and network oscillations in vitro. Trends Neurosci. 2003; 26: 676-682. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/14624852
- Wang X-J, Tegnér J, Constantinidis C, Goldman-Rakic PS. Division of labor among distinct subtypes of inhibitory neurons in a cortical microcircuit of working memory. Proc Natl Acad Sci USA. 2004; 101: 1368-1373. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/14742867
- Katsarou A-M, Moshé SL, Galanopoulou AS. Interneuronopathies and their role in early life epilepsies and neurodevelopmental disorders. Epilepsia Open. 2017; 2: 284-306. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/29062978
- Reid CA, Berkovic SF, Petrou S. Mechanisms of human inherited epilepsies. Prog Neurobiol. 2009; 87: 41-57. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/18952142
- de Lanerolle NC, Kim JH, Robbins RJ, Spencer DD. Hippocampal interneuron loss and plasticity in human temporal lobe epilepsy. Brain Res. 1989; 495: 387-395. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/2569920
- Poduri A, Lowenstein D. Epilepsy genetics—past, present, and future. Curr Opin Genet Dev. 2011; 21: 325-332. PubMed: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4074083/
- Tai C, Abe Y, Westenbroek RE, Scheuer T, Catterall WA. Impaired excitability of somatostatin- and parvalbumin-expressing cortical interneurons in a mouse model of Dravet syndrome. Proc Natl Acad Sci USA. 2014; 111: E3139-E3148. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/25024183
- Ribak C, Bradurne R, Harris A. A preferential loss of GABAergic, symmetric synapses in epileptic foci: a quantitative ultrastructural analysis of monkey neocortex. J Neurosci. 1982; 2: 1725-1735. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/6815309
- Moxon KA, Shahlaie K, Girgis F, Saez I, Kennedy J, et al. From adagio to allegretto: The changing tempo of theta frequencies in epilepsy and its relation to interneuron function. Neurobiol Dis. 2019; 129: 169-181. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/30798003
- Jiruska P, Alvarado-Rojas C, Schevon CA, Staba R, Stacey W, et al. Update on the mechanisms and roles of high-frequency oscillations in seizures and epileptic disorders. Epilepsia. 2017; 58: 1330-1339. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/28681378
- Kann O. The interneuron energy hypothesis: Implications for brain disease. Neurobiol Dis. 2016; 90: 75-85. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26284893
- Ledri M, Madsen MG, Nikitidou L, Kirik D, Kokaia M. Global Optogenetic Activation of Inhibitory Interneurons during Epileptiform Activity. J Neurosci. 2014; 34: 3364-3377. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/24573293
- Ladas TP, Chiang CC, Gonzalez-Reyes LE, Nowak T, Durand DM. Seizure reduction through interneuron-mediated entrainment using low frequency optical stimulation. Exp Neurol. 2015; 269: 120-132. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/25863022
- Balagura G, Iapadre G, Verrotti A, Striano P. Moving beyond sodium valproate: choosing the right anti-epileptic drug in children. Expert Opin Pharmacother. 2019; 1-8. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/31099271
- Franchi SA, Macco R, Astro V, Tonoli D, Savino E, et al. A Method to Culture GABAergic Interneurons Derived from the Medial Ganglionic Eminence. Front Cell Neurosci. 2018; 11: 423. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/29358905
- Corti S, Faravelli I, Cardano M, Conti L. Human pluripotent stem cells as tools for neurodegenerative and neurodevelopmental disease modeling and drug discovery. Expert Opin Drug Discov. 2015; 10: 615-629. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/25891144
- Ding S, Schultz PG. A role for chemistry in stem cell biology. Nat Biotechnol. 2004; 22: 833-840. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15229546
- Ding S, Wu TYH, Brinker A, Peters EC, Hur W, et al. Synthetic small molecules that control stem cell fate. Proc Natl Acad Sci. 2003; 100: 7632-7637. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/12794184
- Watanabe K, Kamiya D, Nishiyama A, Katayama T, Nozaki S, et al. Directed differentiation of telencephalic precursors from embryonic stem cells. Nat Neurosci. 2005; 8: 288-296. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15696161
- Danjo T, Eiraku M, Muguruma K, Watanabe K, Kawada M, et al. Subregional Specification of Embryonic Stem Cell-Derived Ventral Telencephalic Tissues by Timed and Combinatory Treatment with Extrinsic Signals. J Neurosci. 2011; 31: 1919-1933. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/21289201
- Maroof AM, Brown K, Shi SH, Studer L, Anderson SA. Prospective Isolation of Cortical Interneuron Precursors from Mouse Embryonic Stem Cells. J Neurosci. 2010; 30: 4667-4675. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/20357117
- Au E, Ahmed T, Karayannis T, Biswas S, Gan L, et al. A Modular Gain-of-Function Approach to Generate Cortical Interneuron Subtypes from ES Cells. Neuron. 2013; 80: 1145-1158. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/24314726
- Tyson JA, Anderson SA. GABAergic interneuron transplants to study development and treat disease. Trends Neurosci. 2014; 37: 169-177. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/24508416
- Close JL, Yao Z, Levi BP, Miller JA, Bakken TE, et al. Single-Cell Profiling of an In Vitro Model of Human Interneuron Development Reveals Temporal Dynamics of Cell Type Production and Maturation. Neuron. 2017; 96: 949. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/28279351
- Goulburn AL, Stanley EG, Elefanty AG, Anderson SA. Generating GABAergic cerebral cortical interneurons from mouse and human embryonic stem cells. Stem Cell Res. 2012; 8: 416-426. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/22280980
- Chambers SM, Fasano CA, Papapetrou EP, Tomishima M, Sadelain M, et al. Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. Nat Biotechnol. 2009; 27: 275-280.
- Li X-J, Zhang X, Johnson MA, Wang Z-B, LaVaute T, et al. Coordination of sonic hedgehog and Wnt signaling determines ventral and dorsal telencephalic neuron types from human embryonic stem cells. Development. 2009; 136: 4055-4063. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/19906872
- Kim T-G, Yao R, Monnell T, Cho J-H, Vasudevan A, et al. Efficient Specification of Interneurons from Human Pluripotent Stem Cells by Dorsoventral and Rostrocaudal Modulation: Human Interneuron Specification. Stem cells. 2014; 32: 1789-1804. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/24648391
- Maroof AM, Keros S, Tyson JA, Ying S-W, Ganat YM, et al. Directed Differentiation and Functional Maturation of Cortical Interneurons from Human Embryonic Stem Cells. Cell Stem Cell. 2013; 12: 559-572. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/23642365
- Nicholas CR, Chen J, Tang Y, Southwell DG, Chalmers N, et al. Functional Maturation of hPSC-Derived Forebrain Interneurons Requires an Extended Timeline and Mimics Human Neural Development. Cell Stem Cell. 2013; 12: 573-586. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/23642366
- Cunningham M, Cho J-H, Leung A, Savvidis G, Ahn S, et al. hPSC-Derived Maturing GABAergic Interneurons Ameliorate Seizures and Abnormal Behavior in Epileptic Mice. Cell Stem Cell. 2014; 15: 559-573. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/25517465
- Liu Y, Weick JP, Liu H, Krencik R, Zhang X, et al. Medial ganglionic eminence–like cells derived from human embryonic stem cells correct learning and memory deficits. Nat Biotechnol. 2013; 31: 440-447. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/23604284
- Conti L, Pollard SM, Gorba T, Reitano E, Toselli M, et al. Niche-independent symmetrical self-renewal of a mammalian tissue stem cell. PLoS Biol. 2005; 3: e283. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/16086633
- Pollard S, Conti L, Smith A. Exploitation of adherent neural stem cells in basic and applied neurobiology. Regen Med. 2006; 1: 111-118. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/17465825
- Pollard SM, Conti L. Investigating radial glia in vitro. Prog Neurobiol. 2007; 83: 53-67. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/17449166
- Onorati M, Binetti M, Conti L, Camnasio S, Calabrese G, et al. Preservation of positional identity in fetus-derived neural stem (NS) cells from different mouse central nervous system compartments. Cell Mol Life Sci. 2011; 68: 1769-1783. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/20981563
- Goffredo D, Conti L, Di Febo F, Biella G, Tosoni A, et al. Setting the conditions for efficient, robust and reproducible generation of functionally active neurons from adult subventricular zone-derived neural stem cells. Cell Death Differ. 2008; 15: 1847-1856. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/19011641
- Spiliotopoulos D, Goffredo D, Conti L, Di Febo F, Biella G, et al. An optimized experimental strategy for efficient conversion of embryonic stem (ES)-derived mouse neural stem (NS) cells into a nearly homogeneous mature neuronal population. Neurobiol Dis. 2009; 34: 320-331. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/19236914
- Sun Y, Pollard S, Conti L, Toselli M, Biella G, et al. Long-term tripotent differentiation capacity of human neural stem (NS) cells in adherent culture. Mol Cell Neurosci. 2008; 38: 245-258. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/18450476
- Koch P, Opitz T, Steinbeck JA, Ladewig J, Brüstle O. A rosette-type, self-renewing human ES cell-derived neural stem cell with potential for in vitro instruction and synaptic integration. Proc Natl Acad Sci USA. 2009; 106: 3225-3230. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/19218428
- Falk A, Koch P, Kesavan J, Takashima Y, Ladewig J, et al. Capture of neuroepithelial-like stem cells from pluripotent stem cells provides a versatile system for in vitro production of human neurons. PloS One. 2012; 7: e29597. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/22272239
- Colasante G, Lignani G, Rubio A, Medrihan L, Yekhlef L, et al. Rapid Conversion of Fibroblasts into Functional Forebrain GABAergic Interneurons by Direct Genetic Reprogramming. Cell Stem Cell. 2015; 17: 719-734. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26526726
- Sun AX, Yuan Q, Tan S, Xiao Y, Wang D, et al. Direct Induction and Functional Maturation of Forebrain GABAergic Neurons from Human Pluripotent Stem Cells. Cell Rep. 2016; 16: 1942-1953. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/27498872
- Shetty AK, Upadhya D. GABA-ergic cell therapy for epilepsy: Advances, limitations and challenges. Neurosci Biobehav Rev. 2016; 62: 35-47. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26748379
- Upadhya D, Hattiangady B, Shetty GA, Zanirati G, Kodali M, et al. Neural Stem Cell or Human Induced Pluripotent Stem Cell-Derived GABA-ergic Progenitor Cell Grafting in an Animal Model of Chronic Temporal Lobe Epilepsy: Stem Cell Grafting in Chronic Epilepsy Rat Model. Curr Protoc Stem Cell Biol. 2016; 2D.7.1-2D.7.47. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/27532817
- Lee H, Yun S, Kim I-S, Lee I-S, Shin JE, et al. Human Fetal Brain-Derived Neural Stem/Progenitor Cells Grafted into the Adult Epileptic Brain Restrain Seizures in Rat Models of Temporal Lobe Epilepsy. PLoS One. 2014; 9: e104092. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/25105891
- Shetty AK. Hippocampal injury-induced cognitive and mood dysfunction, altered neurogenesis, and epilepsy: Can early neural stem cell grafting intervention provide protection? Epilepsy Behav. 2014; 38: 117-124. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/24433836
- Baraban SC, Southwell DG, Estrada RC, Jones DL, Sebe JY, et al. Reduction of seizures by transplantation of cortical GABAergic interneuron precursors into Kv1.1 mutant mice. Proc Natl Acad Sci. 2009; 106: 15472-15477. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/19706400
- Anderson SA, Baraban SC. Cell Therapy Using GABAergic Neural Progenitors. Jaspers Basic Mech Epilepsies. 2012. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/22787598
- Upadhya D, Hattiangady B, Castro OW, Shuai B, Kodali M, et al. Human induced pluripotent stem cell-derived MGE cell grafting after status epilepticus attenuates chronic epilepsy and comorbidities via synaptic integration. Proc Natl Acad Sci. 2019; 116: 287-296. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/30559206
- Zhu Q, Naegele JR, Chung S. Cortical GABAergic Interneuron/Progenitor Transplantation as a Novel Therapy for Intractable Epilepsy. Front Cell Neurosci. 2018; 12: 167. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/29997478
- Sebe JY, Baraban SC. The promise of an interneuron-based cell therapy for epilepsy. Dev Neurobiol. 2011; 71: 107-117. PubMed: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3059084/
- Hunt RF, Girskis KM, Rubenstein JL, Alvarez-Buylla A, Baraban SC. GABA progenitors grafted into the adult epileptic brain control seizures and abnormal behavior. Nat Neurosci. 2013; 16: 692-697.
- Shibley H, Smith BN. Pilocarpine-induced status epilepticus results in mossy fiber sprouting and spontaneous seizures in C57BL/6 and CD-1 mice. Epilepsy Res. 2002; 49: 109-120. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/12049799
- Hattiangady B, Rao MS, Shetty AK. Grafting of striatal precursor cells into hippocampus shortly after status epilepticus restrains chronic temporal lobe epilepsy. Exp Neurol. 2008; 212: 468-481. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/18579133
- Casalia ML, Howard MA, Baraban SC. Persistent seizure control in epileptic mice transplanted with gamma-aminobutyric acid progenitors. Ann Neurol. 2017; 82: 530-542. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/28833459
- Romariz SAA, Paiva DS, Galindo LT, Barnabé GF, Guedes VA, et al. Medial Ganglionic Eminence Cells Freshly Obtained or Expanded as Neurospheres Show Distinct Cellular and Molecular Properties in Reducing Epileptic Seizures. CNS Neurosci. Ther. 2016; 23: 127-134. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/27770487
- Calcagnotto ME, Ruiz LP, Blanco MM, Santos-Junior JG, Valente MF, et al. Effect of neuronal precursor cells derived from medial ganglionic eminence in an acute epileptic seizure model: Effect of Neuronal Precursor Cells. Epilepsia. 2010; 51: 71-75. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/20618405
- Paiva D de S, Romariz SAA, Valente MF, Moraes LB, Covolan L, et al. Transplantation of inhibitory precursor cells from medial ganglionic eminence produces distinct responses in two different models of acute seizure induction. Epilepsy Behav. 2017; 70: 125-130. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/28427019
Figures:
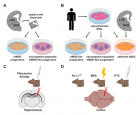
Figure 1
Similar Articles
-
Stem cells in patients with heart failure experienceBenetti Federico*,Natalia Scialacomo. Stem cells in patients with heart failure experience. . 2018 doi: 10.29328/journal.jsctt.1001010; 2: 004-014
-
Preliminary Report on the Effect of Mesenchymal Stem Cell Therapy in Patients with Chronic Lung Allograft DysfunctionCesar A Keller*,Thomas A Gonwa,Athena L Russell,David O Hodge,David B Erasmus,Abba C Zubair. Preliminary Report on the Effect of Mesenchymal Stem Cell Therapy in Patients with Chronic Lung Allograft Dysfunction. . 2018 doi: 10.29328/journal.jsctt.1001012; 2: 035-047
-
Advances in the use of GABAergic interneurons for the treatment of epilepsyGiorgio R Merlo*,Federica Frisina#,Giulia Valetti#,Giulia Zuccarini#,Luciano Conti#. Advances in the use of GABAergic interneurons for the treatment of epilepsy. . 2019 doi: 10.29328/journal.jsctt.1001014; 3: 009-022
-
Clinical Case of Successful Therapy for the Patient with Autism by use of Fetal Stem CellsAA Sinelnyk*, SG Shmyh, IG Matiyashchuk, MO Klunnyk, MP Demchuk, OV Ivankova, OO Honza, IA Susak, MV Skalozub, DV Vatlitsov, KI Sorochynska. Clinical Case of Successful Therapy for the Patient with Autism by use of Fetal Stem Cells. . 2024 doi: 10.29328/journal.jsctt.1001043; 8: 048-053
Recently Viewed
-
Extraction of DNA from face mask recovered from a kidnapping sceneBassey Nsor*,Inuwa HM. Extraction of DNA from face mask recovered from a kidnapping scene. J Forensic Sci Res. 2022: doi: 10.29328/journal.jfsr.1001029; 6: 001-005
-
Sensitivity and Intertextile variance of amylase paper for saliva detectionAlexander Lotozynski*. Sensitivity and Intertextile variance of amylase paper for saliva detection. J Forensic Sci Res. 2020: doi: 10.29328/journal.jfsr.1001017; 4: 001-003
-
The Ketogenic Diet: The Ke(y) - to Success? A Review of Weight Loss, Lipids, and Cardiovascular RiskAngela H Boal*, Christina Kanonidou. The Ketogenic Diet: The Ke(y) - to Success? A Review of Weight Loss, Lipids, and Cardiovascular Risk. J Cardiol Cardiovasc Med. 2024: doi: 10.29328/journal.jccm.1001178; 9: 052-057
-
Could apple cider vinegar be used for health improvement and weight loss?Alexander V Sirotkin*. Could apple cider vinegar be used for health improvement and weight loss?. New Insights Obes Gene Beyond. 2021: doi: 10.29328/journal.niogb.1001016; 5: 014-016
-
Maximizing the Potential of Ketogenic Dieting as a Potent, Safe, Easy-to-Apply and Cost-Effective Anti-Cancer TherapySimeon Ikechukwu Egba*,Daniel Chigbo. Maximizing the Potential of Ketogenic Dieting as a Potent, Safe, Easy-to-Apply and Cost-Effective Anti-Cancer Therapy. Arch Cancer Sci Ther. 2025: doi: 10.29328/journal.acst.1001047; 9: 001-005
Most Viewed
-
Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth EnhancersH Pérez-Aguilar*, M Lacruz-Asaro, F Arán-Ais. Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth Enhancers. J Plant Sci Phytopathol. 2023 doi: 10.29328/journal.jpsp.1001104; 7: 042-047
-
Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case PresentationJulian A Purrinos*, Ramzi Younis. Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case Presentation. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001099; 8: 075-077
-
Feasibility study of magnetic sensing for detecting single-neuron action potentialsDenis Tonini,Kai Wu,Renata Saha,Jian-Ping Wang*. Feasibility study of magnetic sensing for detecting single-neuron action potentials. Ann Biomed Sci Eng. 2022 doi: 10.29328/journal.abse.1001018; 6: 019-029
-
Pediatric Dysgerminoma: Unveiling a Rare Ovarian TumorFaten Limaiem*, Khalil Saffar, Ahmed Halouani. Pediatric Dysgerminoma: Unveiling a Rare Ovarian Tumor. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001087; 8: 010-013
-
Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative reviewKhashayar Maroufi*. Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative review. J Sports Med Ther. 2021 doi: 10.29328/journal.jsmt.1001051; 6: 001-007

HSPI: We're glad you're here. Please click "create a new Query" if you are a new visitor to our website and need further information from us.
If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."